Why Eating Less Isn't Always the Answer: Low Energy Availability and What It Does to Your Body
If you've ever cut calories hard, pushed through fatigue, and still couldn't lose weight — or worse, started feeling worse the longer you dieted — you're not failing. Your body is responding exactly as it was designed to.
The problem isn't your willpower. It's a physiological state called low energy availability (LEA), and it's one of the most common and underrecognized drivers of hormonal dysfunction, metabolic slowdown, and weight loss resistance.
What Is Low Energy Availability?
Energy availability refers to the amount of dietary energy left over for your body's basic physiological functions after accounting for the energy used during physical activity.
In healthy adults, roughly 45 kcal per kilogram of fat-free mass (FFM) per day is considered adequate — approximately equivalent to a true energy balance. When intake falls significantly below this, into the range of below 30 kcal/kg FFM/day in women and below 25 kcal/kg FFM/day in men, the body enters a state of low energy availability and begins making physiological trade-offs to conserve energy (Mountjoy et al., 2023; Papageorgiou et al., 2018).
It's worth noting that much of the early research on LEA was conducted in female athletes, and the hormonal thresholds and timelines can differ between sexes — something we'll come back to. But the core mechanisms are relevant whether you're an athlete or simply someone who has spent months or years on a restrictive diet.
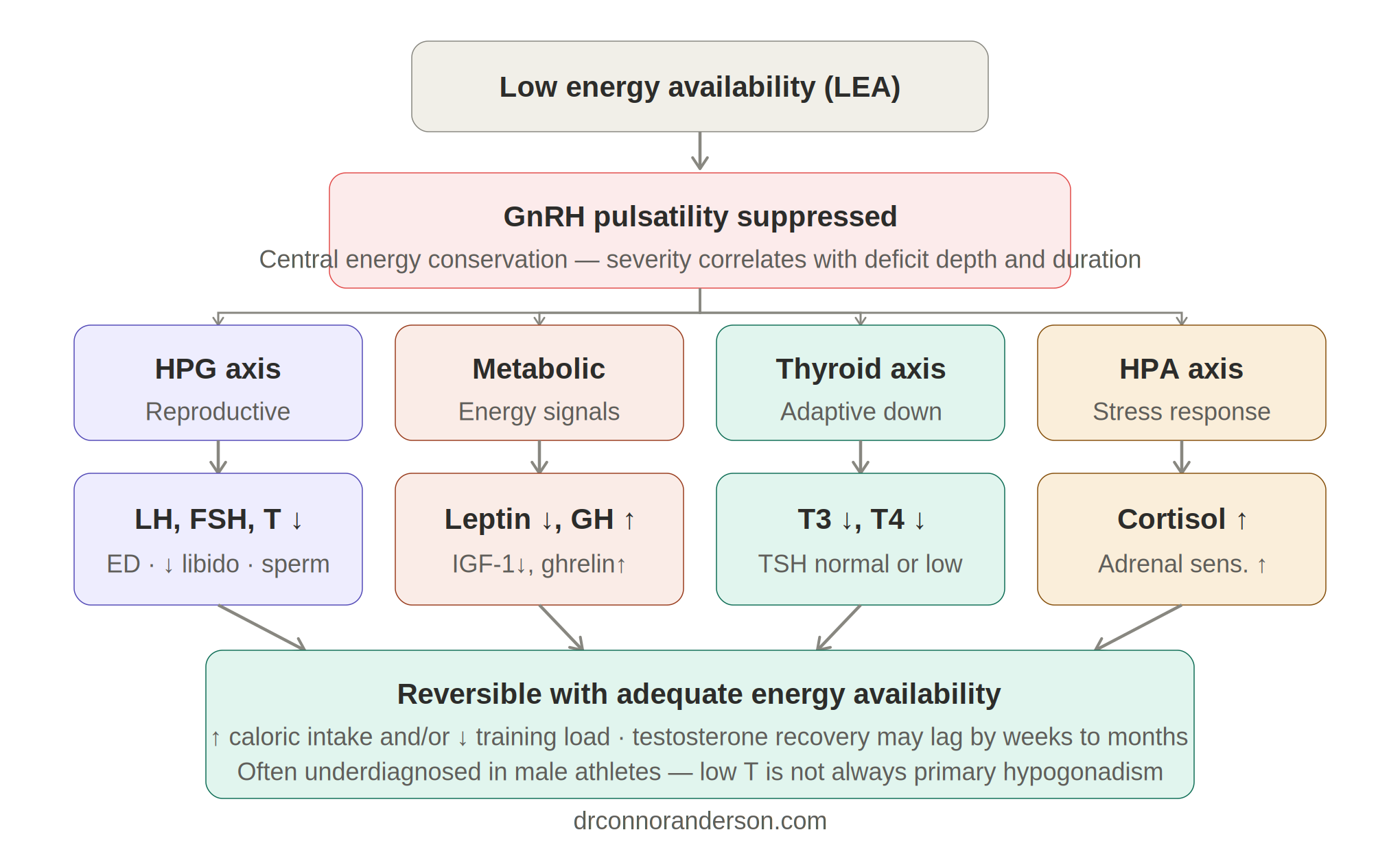
The Cascade: What Happens Inside When You Under-Eat
The Brain Reduces Reproductive Hormone Output
The first thing your body deprioritises under significant energy stress is reproduction. At the centre of this is GnRH (gonadotropin-releasing hormone), produced in the hypothalamus and acting as the master signal for the reproductive hormonal axis. When energy availability is chronically low, GnRH pulsatility is suppressed — and the downstream effects are significant.
Landmark research by Loucks and colleagues established that reducing energy availability below 30 kcal/kg FFM/day suppresses LH pulse frequency, which serves as a proxy for GnRH activity (Loucks & Thuma, 2003). With GnRH suppressed, the pituitary gland reduces output of LH (luteinizing hormone) and FSH (follicle-stimulating hormone) — the hormones that signal the testes to produce testosterone.
In men, the picture is somewhat more nuanced. Short-term, severe energy restriction doesn't always produce immediate testosterone suppression the way it does in women. Instead, notable endocrine disturbances in men — including suppression of testosterone and thyroid hormones — typically occur after sustained exposure to energy availability levels below approximately 20–25 kcal/kg FFM/day, often persisting for several weeks or months (Jeppesen et al., 2025; Fagerberg, 2018, as reviewed in Jeppesen et al., 2025). Observational data in male endurance athletes and cyclists, however, do show meaningful associations between LEA and lower testosterone levels (Cupka & Sedliak, 2023).
Critically, when this occurs, it is not primary hypogonadism (a problem with the testes themselves). It's a functional, centrally-driven suppression — meaning it is reversible when energy intake is restored, though testosterone recovery can lag by weeks to months after energy availability is corrected (Mountjoy et al., 2023).
Your Thyroid Downregulates to Conserve Energy
The thyroid axis is well-documented to respond to caloric restriction. Under conditions of LEA, the body reduces peripheral conversion of T4 into active T3 — the thyroid hormone that primarily drives metabolic rate. Research has shown that even moderate weight loss (around 6%) can produce significant reductions in serum T3, in the absence of meaningful changes in TSH or free T4 (Fontenelle et al., 2014). This means that a standard thyroid panel showing a normal TSH can miss this adaptation entirely.
This T3 decline is driven by reduced energy intake itself, not just weight loss: a randomized controlled trial found that caloric restriction decreased plasma T3 while exercise-induced reductions of similar fat mass did not (Weiss et al., 2008). Carbohydrate availability appears to play a particular role — very low carbohydrate diets have been shown to decrease T3 and increase reverse T3, further impairing active thyroid hormone production (Spaulding et al., 1976, as cited in Weiss et al., 2008).
The result of suppressed T3 is a quieter metabolism: lower body temperature, reduced energy expenditure, fatigue, brain fog, and difficulty losing fat even when eating very little. Many people interpret this fatigue as a sign they need to cut more — which only deepens the problem.
Cortisol Rises
Caloric restriction functions as a physiological stressor. A systematic review and meta-analysis of 27 studies found that short-term low-carbohydrate diets moderately but significantly increased resting cortisol in men (Whittaker & Harris, 2022). In male endurance athletes exposed to LEA, elevated cortisol and a lower testosterone:cortisol ratio have been documented, with more severe and prolonged energy deficits associated with greater cortisol elevation (Harju et al., 2023).
Chronically elevated cortisol promotes muscle tissue breakdown, increases fat storage (particularly around the abdomen), disrupts sleep, and can further suppress testosterone — compounding the hormonal dysfunction set in motion through the HPG axis.
You Feel Constantly Hungry — Because Your Body Is Signalling Correctly
Leptin, a hormone produced by fat cells, suppresses hunger and signals the brain that adequate energy is stored. Under LEA, leptin drops substantially — research has shown decreases of approximately 35% when energy availability falls from 45 to 30 kcal/kg FFM/day, with further reductions of up to 70% at even lower intakes (Loucks & Thuma, 2003). Concurrently, hunger-driving hormones, including ghrelin, rise, and IGF-1 declines.
This produces a state where restriction is simultaneously driving intense hunger signals. This is not a failure of discipline — it is your body doing precisely what it evolved to do when faced with perceived scarcity: maximising drive to restore energy balance.
The Dieting Paradox
Here's what makes LEA so clinically relevant in the context of weight loss: many of the most popular approaches to fat loss — very low-calorie diets, extreme restriction, eliminating entire food groups — are precisely the approaches most likely to trigger this cascade.
A person in chronic LEA will often experience:
Persistent fatigue that doesn't improve with rest
Difficulty losing weight despite eating very little
Low mood, irritability, and poor motivation
Frequent and intense hunger
Reduced libido and sexual function
Feeling cold much of the time
Weight loss plateaus that don't respond to further restriction
What Actually Works: Eating More - Strategically
The research is clear: restoring energy availability is the primary intervention. But it's not simply about eating more of anything. The quality, composition, and distribution of intake matters considerably.
Caloric adequacy: Research and clinical frameworks consistently target ≥45 kcal/kg FFM/day as adequate energy availability, with this value functioning as the benchmark against which LEA is defined (Mountjoy et al., 2023). For most people with a history of aggressive restriction, this represents a meaningful increase.
Carbohydrates matter:Carbohydrate intake is particularly important for thyroid conversion and hypothalamic function. Athletic nutrition guidelines support carbohydrate targets of 5–7 g/kg body weight on training days for individuals doing moderate-to-high training volumes, with lower amounts sufficient for less active individuals (Burke et al., 2011 — IOC Position Stand). The key principle is that chronically restricting carbohydrates in an already under-fuelled person compounds the thyroid and cortisol dysfunction described above, and refeeding with carbohydrates (rather than protein or fat alone) has been shown to normalize T3 levels after restriction (Spaulding et al., 1976).
Protein adequacy: A minimum of ≥1.6 g/kg body weight of protein supports lean mass preservation and recovery, and is consistent with the International Society of Sports Nutrition's position on protein requirements for active individuals (Morton et al., 2018). This also helps buffer the muscle-wasting effects of elevated cortisol during a period of caloric stress.
Whole foods and micronutrient sufficiency: Meeting these targets through whole, minimally processed foods ensures the micronutrient adequacy that synthetic restriction often compromises. Zinc, magnesium, selenium, iodine, vitamin D, and the amino acid tyrosine all play direct roles in testosterone, thyroid, and adrenal function — and are frequently insufficient in highly restricted diets.
Why Extreme Dieting Fails Long-Term
Rigid, highly restrictive diets fail for reasons that are physiological before they are psychological. LEA produces a hormonal profile that is incompatible with sustainable fat loss: suppressed thyroid slows metabolism, elevated cortisol promotes fat storage and muscle loss, and falling leptin intensifies hunger to a point where adherence becomes nearly impossible.
By contrast, an approach that maintains adequate energy availability — using strategic caloric and carbohydrate targets, prioritizing protein, and building from whole foods — keeps the hormonal environment in a state that supports fat loss without triggering the body's conservation response.
Sustainable weight loss is not about eating as little as you can tolerate. It's about eating enough of the right things to keep your metabolism, hormones, and hunger signals working in your favour.
A Note on Testing
Because many of the hormonal changes associated with LEA — particularly thyroid suppression and functional testosterone decline — can appear subtle on standard testing or be missed entirely when only TSH is checked, comprehensive lab work is important for anyone with a history of restrictive dieting or who is experiencing these symptoms.
Useful markers include Total and Free Testosterone, LH, FSH, SHBG, AM cortisol, TSH, fT4, fT3, rTe, IGF-1, vitamin D, zinc, RBC magnesium, and ferritin. Reassessing every 8–12 weeks during a nutrition intervention provides a clear picture of whether the body is responding and allows for targeted adjustments.
This post is for educational purposes and does not constitute medical advice.
Working With Me
These are patterns I have clinical experience working with. My approach helps maintain consistent, realistic, and metabolically healthy weight loss. If you're in Ontario and want to explore what this looks like for you, you can book a consultation here.
Dr. Connor Anderson, ND · Naturopathic Doctor · CONO #4464
References
Agnihothri, R.V., Courville, A.B., Linderman, J.D., Smith, S., Brychta, R., Remaley, A., Chen, K.Y., Simchowitz, L., & Celi, F.S. (2014). Moderate weight loss is sufficient to affect thyroid hormone homeostasis and inhibit its peripheral conversion. Thyroid, 24(1), 19–26. https://doi.org/10.1089/thy.2013.0055
Burke, L.M., Hawley, J.A., Wong, S.H.S., & Jeukendrup, A.E. (2011). Carbohydrates for training and competition. Journal of Sports Sciences, 29(S1), S17–S27. https://doi.org/10.1080/02640414.2011.585473
Cupka, M., & Sedliak, M. (2023). Hungry runners – low energy availability in male endurance athletes and its impact on performance and testosterone: mini-review. European Journal of Translational Myology, 33(2), 11104. https://doi.org/10.4081/ejtm.2023.11104
Jeppesen, J.S., et al. (2025). Short-term severe low energy availability in athletes: Molecular mechanisms, endocrine responses, and performance outcomes — a narrative review. Scandinavian Journal of Medicine & Science in Sports, 35. https://doi.org/10.1111/sms.70089
Koltun, K.J., de Souza, M.J., Scheid, J.L., & Williams, N.I. (2020). Energy availability is associated with luteinizing hormone pulse frequency and induction of luteal phase defects. The Journal of Clinical Endocrinology & Metabolism, 105(1), 185–193. https://doi.org/10.1210/clinem/dgz030
Loucks, A.B., & Thuma, J.R. (2003). Luteinizing hormone pulsatility is disrupted at a threshold of energy availability in regularly menstruating women. The Journal of Clinical Endocrinology & Metabolism, 88(1), 297–311. https://doi.org/10.1210/jc.2002-020369
Morton, R.W., Murphy, K.T., McKellar, S.R., Schoenfeld, B.J., Henselmans, M., Helms, E., Aragon, A.A., Devries, M.C., Banfield, L., Krieger, J.W., & Phillips, S.M. (2018). A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. British Journal of Sports Medicine, 52(6), 376–384. https://doi.org/10.1136/bjsports-2017-097608
Mountjoy, M., Ackerman, K.E., Bailey, D.M., Burke, L.M., Constantini, N., Hackney, A.C., Heikura, I.A., Melin, A., Pensgaard, A.M., Stellingwerff, T., Sundgot-Borgen, J.K., Torstveit, M.K., Jacobsen, A.U., Verhagen, E., Budgett, R., Engebretsen, L., & Erdener, U. (2023). 2023 International Olympic Committee's (IOC) consensus statement on Relative Energy Deficiency in Sport (REDs). British Journal of Sports Medicine, 57(17), 1073–1097. https://doi.org/10.1136/bjsports-2023-106994
Spaulding, S.W., Chopra, I.J., Sherwin, R.S., & Lyall, S.S. (1976). Effect of caloric restriction and dietary composition on serum T3 and reverse T3 in man. The Journal of Clinical Endocrinology & Metabolism, 42(1), 197–200. https://doi.org/10.1210/jcem-42-1-197
Weiss, E.P., et al. (2008). Caloric restriction but not exercise-induced reductions in fat mass decrease plasma triiodothyronine concentrations: a randomized controlled trial. Rejuvenation Research, 11(3), 605–609. https://doi.org/10.1089/rej.2007.0622
Whittaker, J., & Harris, M. (2022). Low-carbohydrate diets and men's cortisol and testosterone: Systematic review and meta-analysis. Nutrition and Health, 28(4), 543–554.